This is a process by which egg cells are fertilised by sperm outside the body, in vitro. IVF is a major treatment in infertility when other methods of assisted reproductive technology have failed. The process involves hormonally controlling the ovulatory process, removing ova (eggs) from the woman’s ovaries and letting sperm fertilise them in a fluid medium. The fertilised egg (zygote) is then transferred to the patient’s uterus with the intent to establish a successful pregnancy.
IVF may be used to overcome female infertility in the woman due to problems of the fallopian tube, making fertilisation in vivo difficult.
For IVF to be successful it typically requires healthy ova, sperm that can fertilise, and a uterus that can maintain a pregnancy. Due to the costs of the procedure, IVF is generally attempted only after less expensive options have failed.
IVF can also be used with egg donation or surrogacy where the woman providing the egg isn’t the same who will carry the pregnancy to term. This means that IVF can be used for females who have already gone through menopause. The donated oocyte can be fertilised in a crucible. If the fertilisation is successful, the embryo will be transferred into the uterus, within which it may implant.
IVF can also be combined with preimplantation genetic diagnosis (PGD) to rule out presence of genetic disorders. A similar but more general test has been developed called Preimplantation Genetic Haplotyping (PGH).
Method
Theoretically, in vitro fertilization could be performed by aspirating contents from a woman’s fallopian tubes or uterus with a plastic catheter after natural ovulation, mix it with semen from a man and reinsert into the uterus. However, without additional techniques, the chances of pregnancy would be extremely small. Such additional techniques that are routinely used in IVF include ovarian hyperstimulation to retrieve multiple eggs, ultrasound-guided transvaginal oocyte retrieval directly from the ovaries, egg and sperm preparation, as well as culture and selection of resultant embryos.
Ovarian hyperstimulation
There are two main protocols for stimulating the ovaries for IVF treatment. The long protocol involves downregulation (suppression or exhaustion) of the pituitary ovarian axis by the prolonged use of a GnRH agonist. Stimulation of the ovaries using a gonadotrophin starts once the process of downregualtion is complete generally after 10 to 14 days.
The short protocol consists of a regimen of fertility medications to stimulate the development of multiple follicles of the ovaries. In most patients, injectable gonadotropins (usually FSH analogues) are used under close monitoring. Such monitoring frequently checks the estradiol level and, by means of gynecologic ultrasonography, follicular growth. Typically approximately 10 days of injections will be necessary. Spontaneous ovulation during the cycle is typically prevented by the use of GnRH agonists that are started prior or at the time of stimulation or GnRH antagonists that are used just during the last days of stimulation; both agents block the natural surge of luteinising hormone (LH) and allow the physician to start the ovulation process by using medication, usually injectable human chorionic gonadotropins. Ovarian stimulation carries the risk of excessive or hyperstimulation. This complication is life threatening and ovarian stimulation using gonadotrophins must only be carried out under strict medical supervision
Transvaginal oocyte retrieval
When follicular maturation is judged to be adequate, human chorionic gonadotropin (hCG) is given. Commonly, this is known as the “trigger shot.”[1] This agent, which acts as an analogue of luteinising hormone, makes the follicles perform their final maturation, and would cause ovulation about 42 hours after injection, but a retrieval procedure takes place just prior to that, in order to recover the egg cells from the ovary. The eggs are retrieved from the patient using a transvaginal technique (transvaginal oocyte retrieval) involving an ultrasound-guided needle piercing the vaginal wall to reach the ovaries. Through this needle follicles can be aspirated, and the follicular fluid is handed to the IVF laboratory to identify ova. It is common to remove between ten and thirty eggs. The retrieval procedure takes about 20 minutes and is usually done under conscious sedation or general anaesthesia.
Egg and sperm preparation
In the laboratory, the identified eggs are stripped of surrounding cells and prepared for fertilisation. An oocyte selection may be performed prior to fertilisation to select eggs with optimial chances of successful pregnancy. In the meantime, semen is prepared for fertilisation by removing inactive cells and seminal fluid in a process called sperm washing. If semen is being provided by a sperm donor, it will usually have been prepared for treatment before being frozen and quarantined, and it will be thawed ready for use.
Fertilisation
The sperm and the egg are incubated together at a ratio of about 75,000:1 in the culture media for about 18 hours. A single sperm may be injected directly into the egg using intracytoplasmic sperm injection (ICSI). The fertilised egg is passed to a special growth medium and left for about 48 hours until the egg consists of six to eight cells.
Gamete intrafallopian transfer, eggs are removed from the woman and placed in one of the fallopian tubes, along with the man’s sperm. This allows fertilisation to take place inside the woman’s body. Therefore, this variation is actually an in vivo fertilisation, not an in vitro fertilisation.
Embryo culture
Typically, embryos are cultured until having reached the 6–8 cell stage three days after retrieval. In many Canadian, American and Australian programmes, however, embryos are placed into an extended culture system with a transfer done at the blastocyst stage at around five days after retrieval, especially if many good-quality embryos are still available on day 3. Blastocyst stage transfers have been shown to result in higher pregnancy rates in Europe, transfers after 2 days are common.
Culture of embryos can either be performed in an artificial culture medium or in an autologous endometrial coculture (on top of a layer of cells from the woman’s own uterine lining). With artificial culture medium, there can either be the same culture medium throughout the period, or a sequential system can be used, in which the embryo is sequentially placed in different media. For example, when culturing to the blastocyst stage, one medium may be used for culture to day, and a second medium is used for culture thereafter.Single or sequential medium are equally effective for the culture of human embryos to the blastocyst stage. Artificial embryo culture media basically contain glucose, pyruvate, and energy-providing components, but addition of amino acids, nucleotides, vitamins, and cholesterol improve the performance of embryonic growth and development.
Embryo selection
Laboratories have developed grading methods to judge oocyte and embryo quality. In order to optimise pregnancy rates. Preimplantation genetic diagnosis (PGD) or screening may be performed prior to transfer in order to avoid inheritable diseases
Embryo transfer
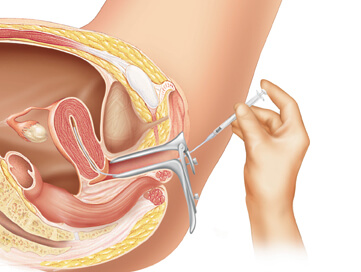
Embryos are graded by the embryologist based on the number of cells, evenness of growth and degree of fragmentation. The number to be transferred depends on the number available, the age of the woman and other health and diagnostic factors. The embryos judged to be the “best” are transferred to the patient’s uterus through a thin, plastic catheter, which goes through her vagina and cervix. Several embryos may be passed into the uterus to improve chances of implantation and pregnancy.
Pregnancy rates
Pregnancy rate is the success rate for pregnancy. For IVF, it is the percentage of all attempts that lead to pregnancy, which generally refers to treatment cycles where eggs are retrieved and fertilised in vitro. Pregnancies that are delivered with a viable baby are called live birth rate.
Because not each IVF cycle that is started will lead to oocyte retrieval or embryo transfer, reports of live birth rates need to specify the denominator, namely IVF cycles started, IVF retrievals, or embryo transfers. The Society for Assisted Reproductive Technology (SART) summarised 2008-9 success rates for US clinics for fresh embryo cycles that did not involve donor eggs and gave live birth rates by the age of the prospective mother, with a peak at 41.3% per cycle started and 47.3% per embryo transfer for patients under 35 years of age.
IVF attempts in multiple cycles result in increased cumulative live birth rates. Depending on the demographic group, one study reported 45% to 53% for three attempts, and 51% to 71% to 80% for six attempts.
Success or failure factors
The main potential factors that influence pregnancy (and live birth) rates in IVF have been suggested to be maternal age, duration of infertility or subfertility, bFSH and number of oocytes, all reflecting ovarian function. Optimal woman’s age is 23–39 years at time of treatment
Other factors
Other determinants of outcome of IVF include:
- Tobacco smoking reduces the chances of IVF producing a live birth by 34% and increases the risk of an IVF pregnancy miscarrying by 30%.
- A body mass index (BMI) over 27 causes a 33% decrease in likelihood to have a live birth after the first cycle of IVF, compared to those with a BMI between 20 and 27 Also, pregnant women who are obese have higher rates of congenital abnormality, miscarriage, gestational diabetes, hypertension, thromboembolism and problems during delivery. Ideal body mass index is 19–30.
- Salpingectomy before IVF treatment increases chances for women with hydrosalpinges
- Success with previous pregnancy and/or live birth increases chances
- Low alcohol/caffeine intake increases success rate
- The number of embryos transferred in the treatment cycle
- Other factors of semen quality for the sperm provider.
Risks
During ovarian stimulation, hyperstimulation syndrome may occur. This results in swollen, painful ovaries and some form of it (mild, moderate or severe) occurs in 30% of patients. Mild cases can be treated with over the counter meds and cases can be resolved in the absence of pregnancy. In moderate cases, ovaries swell and fluid accumulated in the abdominal cavities and may have symptoms of heartburn, gas, nausea or loss of appetite. In severe cases patients have sudden excess abdominal pain, nausea, vomiting and will result in hospitalization.
During egg retrieval, there’s a small chance of bleeding, infection, and damage to surrounding structures like bowel and bladder (transvaginal ultrasound aspiration) as well as difficulty breathing, chest infection, allergic reactions to meds, or nerve damage (laproscopy). During embryo transfer, if more than one embryo is transferred there’s always a risk of multiple pregnancy, infertile couples may see this is good news but there may be risk to the embryos and to the mother such as premature delivery. Ectopic Pregnancy may also occur- fertilized egg develops outside the uterus, usually in the fallopian tubes and requires immediate destruction of the fetus.
Complications
The major complication of IVF is the risk of multiple births. This is directly related to the practice of transferring multiple embryos at embryo transfer. Multiple births are related to increased risk of pregnancy loss, obstetrical complications, prematurity, and neonatal morbidity with the potential for long term damage
Another risk of ovarian stimulation is the development of ovarian hyperstimulation syndrome, particularly if hCG is used to “trigger ovulation”.